The Clot Retriever Device obtained medical device registration certificate
In September 2020, the Clot Retriever Device independently developed by Zhuhai TonBridge Medical Technology Co., Ltd. obtained the medical device registration certificate approved by NMPA.
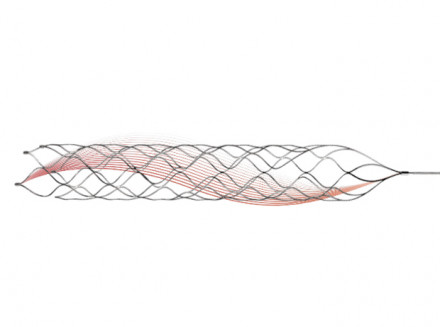
The Clot Retriever Device is used for the treatment of ischemic stroke. It has a unique open structure with spiraling sides, which can rotate and entangle the thrombus, resulting in a stronger clamping force on the thrombus.
If you want to know more information, please sent a message to tonbridge medical group email.
We are one of the leading companies in the peripheral and neurovascular interventional medical device market in China. As a fully integrated medical device company, zylox tonbridge medical has strong R&D and manufacturing capabilities, multiple specialized technology platforms, and proven commercialization capabilities. Led by our talented management team, we strive to provide best medical services and cutting-edge technical solutions to physicians and patients in China and around the world.
We can also offer kinds of venous closure device for sale. If you are interested, please contact us.